Fighting Coronavirus Together: WHO Announces Pause of Hydroxychloroquine Drug Trial in Search for COVID-19 Cure
KIU, Main Campus – On the World Africa Day, 25th May 2020, the World Health Organization (WHO) held a media briefing to update the public on the COVID-19 pandemic.
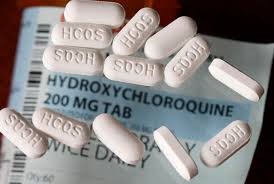
A group of participants in the WHO's Solidarity Trial, which aims to find a treatment for COVID-19, agreed to review all available evidence resulting from an observational study carried out by The Lancet - one of the world's oldest and best-known weekly peer-reviewed general medical journals - on the controversial drugs hydroxychloroquine and chloroquine, namely on their effects on hospital patients suffering from the Coronavirus. This study estimated a "higher mortality rate" among patients receiving the drug, with the risk of death increasing by 34% and serious heart arrhythmias by 137%.
The WHO has, therefore, announced a "temporary pause" of the hydroxychloroquine section of the trial while the data is reviewed by the Data Safety Monitoring Board.
"I wish to reiterate that these drugs are accepted as generally safe for use in patients with autoimmune diseases or malaria," said WHO Director-General, Dr Tedros Adhanom Ghebreyesus.
Picture credit: Internet photo

 Rebecca Nabejja
Rebecca Nabejja
