
Apr
Bunyoro-Tooro Students Association Participate in Games to Mark Their Sports Day
April 23, 2024, 8:32 am
 Collins Kakwezi
Collins Kakwezi
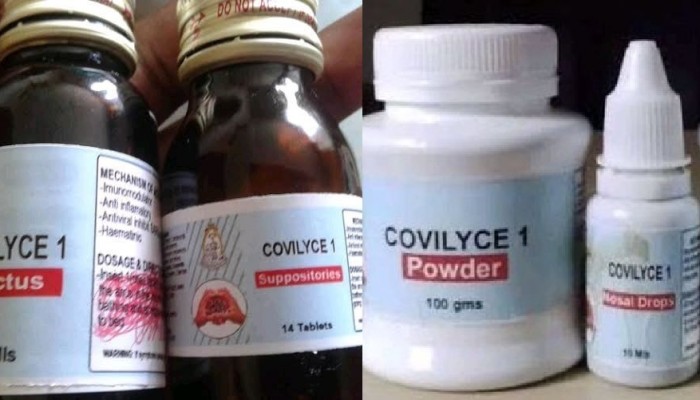
The National Drug Authority (NDA) has ordered Gulu University to cease production of Covilyce-1, a herbal medicine that the varsity developed to treat COVID-19, saying it has not yet been approved, according to local newspaper Daily Monitor.
The statutory regulator’s directive comes a few days after the university scientists led by Dr. Alice Veronica Lamwaka announced that 100 COVID-19 patients who used Covilyce-1 had recovered.
Dr. Lamwaka reportedly made an appeal for Shs200m support to increase production due to soaring demand for the herbal medicine several of whose users testify that it works to treat the pandemic.
Nevertheless, Abiaz Rwamwiri, the NDA spokesman, told Daily Monitor that the Authority had dispatched a team of its experts to northern Uganda to stop Gulu University from producing the medicine, citing safety and efficacy concerns.
Image: The Pearl Times
Kampala International University,
Box 20000, Ggaba Road, Kansanga, Kampala