
Sep
KIU Teaching Hospital Strengthens Partnership With Kitagata General Hospital
September 5, 2024, 9:40 am
 Administrator
Administrator
Hepatitis B virus (HBV) is one of the major public health concerns globally. It is highly infectious and can be transmitted from person to person through vertically or horizontally via contaminated body fluids. Despite the provision of an effective vaccine, it remains a major problem worldwide, particularly among the developing countries.
HBV is a life-threatening infection which attacks liver cells and can cause acute and chronic disease. The acute HBV infection is usually a self-limiting disease; however, chronic infection can cause liver cancer that becomes the second most common cause of death among cancer disease globally.
Annually more than 686,000 people die related to HBV infection complications. Though HBV vaccination is the most important prevention mechanism, it is less likely accessible for private access due to the high cost of the vaccine.
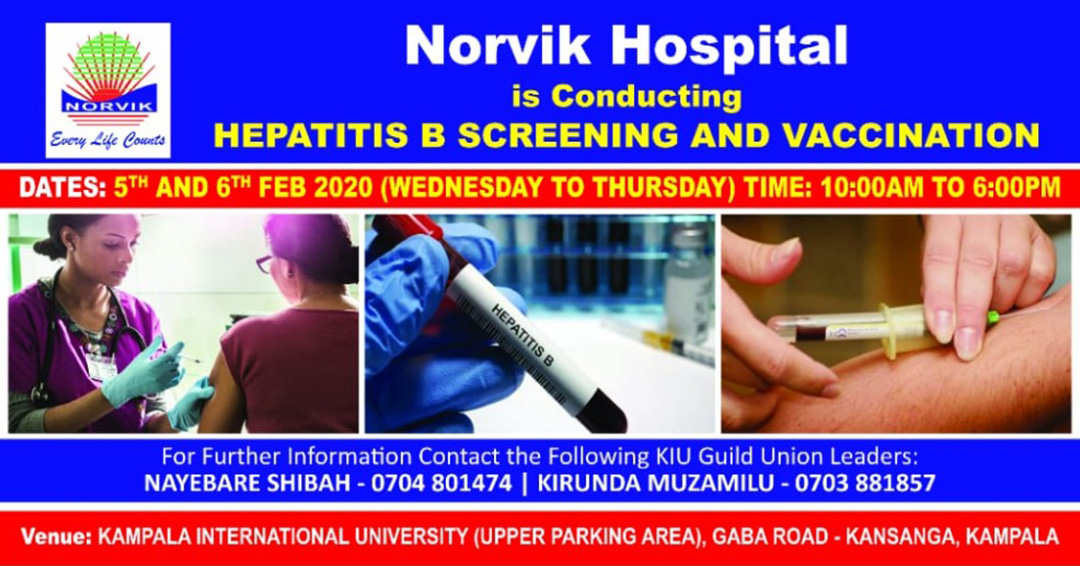
Therefore, in a bid to respond to the above health burden, Norvik Hospital is set to conduct Hepatitis B screening and vaccination on the 5th and 6th of February 2020 at the KIU Main Campus. The services will be provided from 10:00 am to 6:00 pm by health workers from Norvik Hospital through their outreach program. The main objective of this program is to increase awareness and coverage of Hepatitis B screening and vaccination.
In this process, the students will be followed up to receive all three doses that are, the first and the second dose after one month, then the third dose after 5 months.
Transmission
Hepatitis B is also spread by needle stick injury, tattooing, piercing, and exposure to infected blood and body fluids, such as saliva and, menstrual, vaginal, and seminal fluids. Sexual transmission of Hepatitis B may occur, particularly in unvaccinated individuals engaging in sexual activity who have sex with multiple sex partners or contact with sex workers.
Infection in adulthood leads to chronic hepatitis in less than 5% of cases, whereas infection in infancy and early childhood leads to chronic hepatitis in about 95% of cases. Transmission of the virus may also occur through the reuse of needles and syringes either in health-care settings or among persons who inject drugs. In addition, the infection can occur during medical, surgical and dental procedures, through tattooing, or through the use of razors and similar objects that are contaminated with infected blood.
The Hepatitis B virus can survive outside the body for at least 7 days. During this time, the virus can still cause infection if it enters the body of a person who is not protected by the vaccine. The incubation period of the Hepatitis B virus is 75 days on average but can vary from 30 to 180 days. The virus may be detected within 30 to 60 days after infection and can persist and develop into chronic hepatitis B.
Symptoms
Most people do not experience any symptoms when newly infected. However, some people have acute illnesses with symptoms that last several weeks, including yellowing of the skin and eyes (jaundice), dark urine, extreme fatigue, nausea, vomiting, and abdominal pain. A small subset of persons with acute hepatitis can develop acute liver failure, which can lead to death.
In some people, the HBV can also cause a chronic liver infection that can later develop into cirrhosis (a scarring of the liver) or liver cancer.
Who is at risk of chronic disease?
The likelihood that infection becomes chronic depends on the age at which a person becomes infected. Children less than 6 years of age who become infected with the hepatitis B virus are the most likely to develop chronic infections.
In infants and children:
c�c 80c��??90% of infants infected during the first year of life develop chronic infections; and
c�c 30c��??50% of children infected before the age of 6 years develop chronic infections.
In adults:
c�c less than 5% of otherwise healthy persons who are infected as adults will develop chronic infections; and
c�c 20c��??30% of adults who are chronically infected will develop cirrhosis and/or liver cancer.
Prevention
The Hepatitis B vaccine is the backbone of Hepatitis B prevention. The World Health Organization (WHO) recommends that all infants receive the Hepatitis B vaccine as soon as possible after birth, preferably within 24 hours. In most cases, 1 of the following 2 options is considered appropriate:
c�c a 3-dose schedule of Hepatitis B vaccine, with the first dose (monovalent) given at birth and the second and third doses (monovalent or combined vaccine) given at the same time as the first and third doses of diphtheria, pertussis (whooping cough), and tetanus c��?? (DTP vaccine); or
c�c a 4-dose schedule, where a monovalent birth dose is followed by 3 monovalent or combined vaccine doses, usually given with other routine infant vaccines.
The complete vaccine series induces protective antibody levels in more than 95% of infants, children and young adults. Protection lasts at least 20 years and is probably lifelong. Thus, WHO does not recommend booster vaccinations for persons who have completed the 3-dose vaccination schedule.
All children and adolescents younger than 18 years and not previously vaccinated should receive the vaccine especially in settings where it is possible that more people in high-risk groups may acquire the infection and should thereby be vaccinated. This includes:
c�c people who frequently require blood or blood products, dialysis patients and recipients of solid organ transplantations;
c�c people in prisons;
c�c people who inject drugs;
c�c household and sexual contacts of people with chronic HBV infection;
c�c people with multiple sexual partners;
c�c healthcare workers and others who may be exposed to blood and blood products through their work; and
c�c travelers who have not completed their HBV series, who should be offered the vaccine before leaving for endemic areas.
The vaccine has an excellent record of safety and effectiveness. Since 1982, over 1 billion doses of Hepatitis B vaccine have been used worldwide. In many countries where 8c��??15% of children used to become chronically infected with the HBV, vaccination has reduced the rate of chronic infection to less than 1% among immunized children.
Kampala International University,
Box 20000, Ggaba Road, Kansanga, Kampala
+256-760 502660
+256-700 100808